New RNA Telomere Therapy Reverses Aging
Summary: Doctors lengthen telomeres with RNA therapy to reverse aging in human cells, according to a new research report. Telomere attrition is one of the nine hallmarks of aging. [Author: Brady Hartman] This article first appeared on LongevityFacts.]
Dr. John Cooke is department chair of cardiovascular sciences at Houston Methodist Research Institute and is the lead author of a recent paper published in the Journal of the American College of Cardiology. Dr. Cooke’s team used RNA therapy to lengthen the telomeres of patients’ cells, making them younger in the process. In a video statement accompanying the report, the lead author remarked:
“We can make aged cells younger.”
How Cooke Lengthens Telomeres
Dr. Cooke’s team took cells from children with a disease called progeria, a rare genetic condition that causes them to age rapidly. Cooke said that 12 out of the 17 participants in the study already had shortened telomeres, which appeared similar to aging cells from an elderly man. Dr. Cooke suspected that if he could lengthen the telomeres of the cells, he could improve their health.
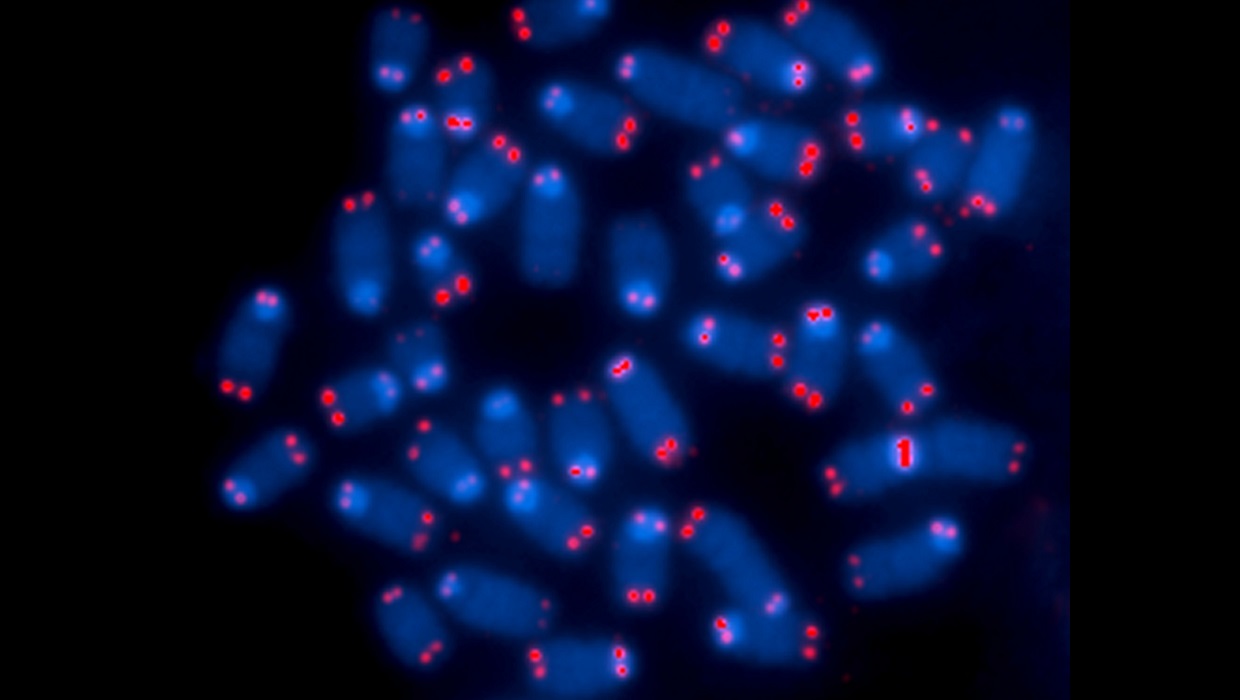
RNA Therapy Lengthens Telomeres
Cooke’s team used a new technology called RNA therapeutics, a novel procedure that inserts RNA directly into cells. As soon as it was inserted, the foreign RNA spurred the cells to produce telomerase, a molecule that lengthens telomeres.
Telomeres protect the ends of chromosomes keeping the DNA neat and tidy. The novel procedure lengthened the telomeres of the cells and improved their function and lifespan.
The lead author noted that before the RNA therapy, the cells would multiply poorly and die quickly. After inserting the therapeutic RNA, Dr. Cooke said that the “cells proliferated normally. It was a dramatic improvement.“ The lead author also noted that markers of aging, such as inflammatory proteins, were also reduced.
Excited by these promising results with human cell cultures, Dr. Cooke plans to do more advanced research with animals, and if that proves successful, the next step is human testing. The researcher believes that RNA therapy can fight the chronic diseases of aging and maybe reverse aging itself. The lead author said,
“When we lengthen telomeres, that can reverse a lot of the problems associated with aging.”
The novel RNA therapy earns a place alongside promising yet unproven life-extending treatments, such as parabiosis – also called young blood old blood – a technique used to transfer anti-aging molecules and stem cells to older individuals.
While a promising anti-aging intervention, telomere therapy hasn’t yet reached the status of the leading lifespan-extending treatments such as the anti-aging drug metformin; and the most famous lifespan-extending of them all, rapamycin.
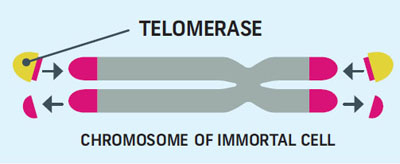
How Telomerase Lengthens Telomeres
What is a Telomere?
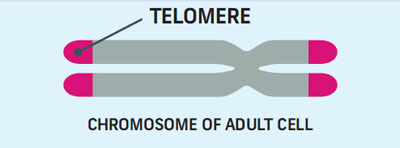
Like the plastic caps at the end of your shoelaces, telomeres are a protective cap on the ends of chromosomes. Every time a cell divides, those protective caps shorten. The older you get, the shorter your telomeres get. Once a cell has divided about 50 times, it’s time for the cell to self-destruct.
Telomerase Lengthens Telomeres
Many different types of proteins are involved in telomere up-keep. The most notable of which is telomerase, a compound which rebuilds the shortened ends. The rank and file cells of the body don’t use telomerase, while immortal cells do.
Embryo cells and stem cells are involved in growth and regeneration and need to be immortal to do their jobs. They don’t like being limited, so they add back their telomere units, using telomerase. This way, they keep their telomere length the same despite repeated cell divisions. Unfortunately, cancer cells use telomerase as well. They also keep their telomere length constant and remain immortal, until they kill their host.
Life Without Telomerase

Rank and file cells are prohibited from using telomerase, and their telomeres shorten with each division until they die. When all a cell has divided about 50 times and all the telomeres have been removed, the cell usually self-destructs, in a process call apoptosis.
Cellular Senescence – Turning To The Dark Side

Some cells, however, refuse to self-destruct and become senescent cells. Senescent cells produce oodles of inflammatory compounds and cause a great deal of trouble throughout the body. Fortunately, researchers are developing anti-aging senolytic drugs, compounds which rid the body of worn-out senescent cells.
Telomere Length And Lifespan
Telomere length is indirectly proportional to lifespan. While it doesn’t make intuitive sense, the fact of the matter is that long telomeres are associated with short lifespans and short telomeres with shorter ones.
Paradoxically, as an organism ages, it’s average telomere length decreases.
Is Longer Better?
Should we lengthen telomeres to live longer, healthier lives?
Scientists are divided on the answer. One group argues that using telomerase or RNA therapy to lengthen telomeres can eliminate the chronic diseases of aging, from type 2 diabetes to cancer.
The other side cautions that because cancer cells use telomerase to make themselves immortal, that adding more telomerase to our bodies will riddle our bodies with cancer.
Can We Lengthen Telomeres To Stop Aging?
Researchers believe that shortening of the telomeres leads to the aging of cells and of the whole organism. Scientists also believe that the telomere mechanism evolved to prevent cells replicating infinitely. Unfortunately, the telomere mechanism also makes cancer possible.
Telomeres shorten with age, and that produces senescent cells, a significant source of inflammation and the chronic diseases of aging. In theory, giving drugs that stimulate additional telomerase could lengthen telomeres, which in turn prevents the degenerative processes that accompanying aging such as heart disease, cancer, type 2 diabetes, and arthritis.
However, scientists fear that it may also lead to cancer. As well, Dr. Cooke’s research showed that extending the telomere length improved the health and lifespan of the cells. The caveat is that the children’s cells had abnormally short telomeres, to begin with, due to being afflicted with progeria. Shorter telomeres cause genomic instability and may be a risk factor for developing cancer.
Bottom Line
Scientists have been intrigued by the observation that as people’s telomeres get shorter, their risk of death and of many chronic diseases of aging also rises. So one would think that any drug that rebuilds or lengthens telomeres would be an instant blockbuster. Unfortunately, researchers have observed that chemically pushing telomerase enzymes to lengthen telomeres tend to make cells cancerous.
Telomerase therapy has the potential to be used in rejuvenation and regeneration applications. The best use of this therapy is in regenerative medicine. As Dr. Cooke’s experiments on cell cultures showed, inserting telomerase into dysfunctional cells can lengthen telomeres, making the cells healthier.
Help Us Spread the Word
Please share this post and help us spread the word. All it takes is one simple click on any of the social media links shown on this page.
References / Additional Reading on Telomeres
Yanhui Li, Gang Zhou, Ivone G. Bruno, John P. Cooke. Telomerase mRNA Reverses Senescence in Progeria Cells. Journal of the American College of Cardiology. 2017. Volume 70, Issue 6, Pages 804-805. Available Online.
P. Scaffidi, L. Gordon, T. Misteli. The Cell Nucleus and Aging: Tantalizing Clues and Hopeful Promises. PLoS Biology Vol. 3/11/2005, e395 doi:10.1371/journal.pbio.0030395. Available Online.
De Jesus, E. Vera, K. Schneeberger, AM Tejera, E. Ayuso, F. Bosch, & MA Blasco. Telomerase gene therapy in adult and old mice delays aging and increases longevity without increasing cancer. August 2012, EMBO Molecular Medicine. doi:10.1002/emmm.201200245. Available Online.
Disclaimer
Diagnosis, Advice, and Treatment: This article is intended for informational and educational purposes only and is not a substitute for professional medical advice. RNA therapy and other compounds which lengthen telomeres are experimental techniques which have not been proven to be safe and effective in clinical trials. Experimental drugs and treatments carry a much higher risk than FDA-approved ones. The information provided in this report should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. Consult a licensed physician for the diagnosis and treatment of any and all medical conditions. Call 911, or the equivalent emergency hotline number, for all medical emergencies. As well, consult a licensed physician before changing your diet, supplement or exercise programs. Photos, External Links & Endorsements: This article is not intended to endorse companies, organizations or products. Links to external websites, depiction/mention of company names or brands, are intended only for illustration and do not constitute endorsements.
You need to fix the pics .. nice article .. well written ..
thanks
When will they do the clinical trials?
Additional question – When will this be available?