Revolutionary Geroscience Field Targets Aging
Summary: How the field of geroscience aims to slow down the chronic diseases of aging. Part 3 of the 4-part essay ” Geroscience ” by Felipe Sierra. [With an introduction by Brady Hartman. ]
Scientists in the geroscience field aim to slow down the chronic diseases of aging. They have already significantly slowed down aging in laboratory animals and hope to do the same thing for humans.
This article is a continuation of the essay Geroscience by Felipe Sierra, Ph.D. In this 3rd of 4 segments, also titled Geroscience, Dr. Sierra shows us how longevity researchers, called geroscientists, are trying to slow the chronic diseases of aging by understanding and delaying the underlying biological processes of aging.
Felipe Sierra, Ph.D. is the Director of the Division of Aging Biology at the National Institute on Aging (NIA) and is leading the U.S. government’s geroscience research efforts. The NIA is looking into why we age and searching for ways to slow the process down.
Key Points of this Section
- Aging is the major risk factor for most chronic diseases of aging that affect older adults.
- The chronic diseases of aging include arthritis, cancer, sarcopenia (frailty), immune system decline, chronic respiratory diseases such as COPD, macular degeneration, type 2 diabetes, kidney disease, heart disease, vascular disease, hearing decline, and neurodegenerative disorders, such as Alzheimer’s disease and Parkinson’s.
- Researchers have identified a finite number of factors that control the rate of aging at the molecular and cellular levels.
- Scientists can slow the rate of the biologic aging in lab animals using a variety of behavioral, genetic, and pharmacologic interventions.
- When scientists use these interventions to slow the rate of aging, the slowdown is most often accompanied by a delay in and decrease of the severity of naturally occurring diseases and conditions as well as improved resistance to laboratory-induced diseases.
GEROSCIENCE
(from the article Geroscience by Felipe Sierra, Ph.D.
Epidemiologic studies suggest that aging might be the major risk factor for most age-related chronic diseases, [97] and much recent research aimed primarily at increasing life span has shown that this leads to a delay and softening of the diseases that normally accompany old age. [44,98,99] It should be emphasized that the goal of aging biology research is not to extend life span, but to increase health in later years, termed health span. Life span is merely used as a surrogate that is easier to measure, especially in genetically tractable low organisms such as C. elegans, Drosophila melanogaster, and yeast. The close relationship between aging biology and risk for age-related chronic diseases gave rise to the new field of geroscience, “an interdisciplinary field that aims to understand the relationship between aging and age-related diseases and disabilities” [15,100] (also see http://en.wikipedia.org/wiki/Geroscience; [link to article] Figure 7-1). That aging biology plays a role in age-related diseases is not a new concept. There is a wide consensus that the ultimate goal of biomedical research is to improve the quality of human life.
Initial efforts were successful in eradicating or limiting the ravages of infectious diseases and, together with improvements in sanitation and public health, led to a dramatic increase in human longevity during the last century. Unfortunately, that triumph came with a price tag, because it is now known that chronic diseases and conditions of older adults represent the main hurdle toward reaching the goal of improving the quality of human life. Thus, the basic tenet of geroscience is that because aging is malleable, at least in many animal models, and aging is also the main risk factor for most diseases and conditions affecting humans, then addressing the basic biology of aging is likely to return a better payoff in terms of health than addressing diseases one at a time, as often done currently.
Conceptually, it is important to understand that unlike infectious diseases or genetic disorders, chronic diseases of aging are multifactorial and complex in nature. The current model used in biomedicine is focused on fighting disease-specific risk factors for different diseases (e.g., cholesterol for cardiovascular disease, glucose homeostasis for diabetes, amyloid deposits for Alzheimer disease). However, there is a growing realization that although important, those disease-specific factors are not sufficient, and overt disease often becomes apparent only after other elements are present, notably the environment and the receptive niche provided by aging itself [101] (Figure 7-2). For example, unless genetically driven, cancer often appears in humans in their 60s and 70s; the explanation often given is that it takes a long time for all the necessary mutations to accumulate in any given cell. However, most mice develop cancer by the age of 2 years. Mutation and repair rates are roughly comparable between mice and humans, [102-105] and the commonality is that in both cases, cancer strikes when individuals are well past midlife, when aging organisms become frail and lose resilience. A separate issue that makes geroscience timely is the inadequacy of current efforts at addressing one disease at a time.
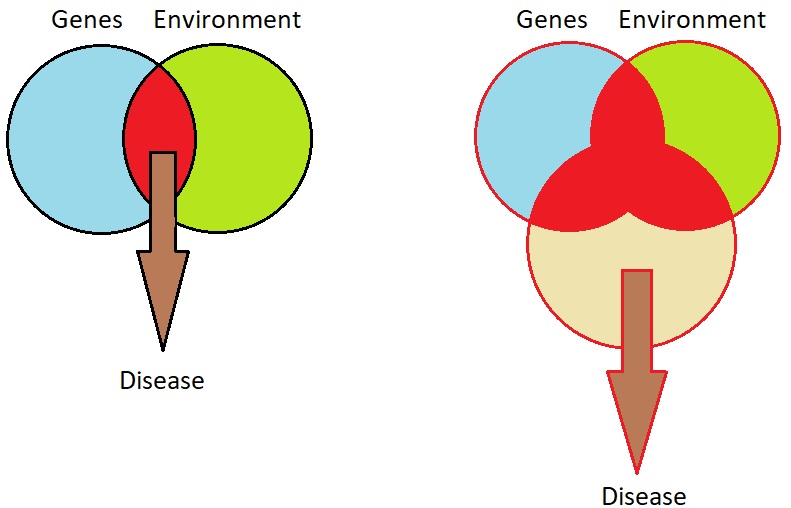
Figure 7-2. The major risk factors for disease. Disease can have multiple causes, grouped primarily into genes, environment, and aging and their interactions. A, When an individual is young, by definition, aging plays no role, and thus disease risk is determined solely by genetics, environment, and gene × environment interactions (shown by the red-margined intersection). B, In contrast, as an individual ages, the role of aging as risk factor becomes ever more apparent until, at extreme age, it is the predominant risk factor for disease. Note that as this happens, the overall risk (the intersection containing at least two risk factors) becomes larger, which explains the age-related increase in overall morbidity. Centenarians are also subject to these variables, but appear to have a lower risk for disease as a result of a cohort effect—all other members of the cohort are dead, and survivors are those who are most resilient.
However, this is seldom seen in older adults, with comorbidities being the norm rather than the exception. Much current research in biomedicine is focused on preventing, curing, or managing one disease at a time, which is reflected at the level of the clinic, academia, and even funding agencies, such as the National Institutes of Health (NIH). This focus has further adverse effects; for example, clinical trials often exclude patients suffering from diseases other than the one being examined and often have an upper limit for age recruitment, thus effectively eliminating the very population for which the intervention is being tested from the trials—comorbid older adults. Because aging is the major risk factor for most chronic conditions, it follows logically that comorbidities are probably the direct result of that. Thus, by embracing that concept, geroscience aims at putting the emphasis on preventing or curing not one but all chronic diseases at once.
It has been known for centuries that aging is the major risk factor for disease and morbidity but is it true that nothing can be done about it? This is a major fallacy that has also been known to be incorrect for centuries. Chronologic age—the number of years humans have been on the globe—cannot be changed, but not everyone ages at the same rate, and there are large differences in the physiologic age of individuals in the same cohort. These differences are commonly believed to be driven by genetics and the environment so that in general, people who exercise, eat moderately, have a balanced diet, and avoid excessive stress tend to have a better physiologic makeup, even in their old age (although some “randomness” also plays a role). Such observations imply that the process of aging is not immutable but, on the contrary, is intrinsically malleable. In laboratory animals ranging from yeast to mice, aging can be delayed by behavioral (diet restriction), [2,3] genetic (more than 700 genes have been described that lead to extended life span), [13] and pharmacologic means (e.g., rapamycin, acarbose, metformin, and even resveratrol, under metabolically stressed conditions). [44,106-109] The significant advances in understanding the basic biology of aging that have been discussed indicate that the field is poised for further discoveries in the realm of pharmacologic means of extending life span. In most cases, these manipulations lead to significant resistance to disease and improvements in physiology; that is, the interventions also increase health span. Most people, including older adults, do not consider extending life span to be a worthy goal unless accompanied by at least an extension of health span and, ideally, a compression of morbidity. As noted, this is also the goal of aging biology research and geroscience. Life span is simply used in research as an easy to measure binary surrogate. The issue of health is much more complex and should never be equated with absence of disease. The increased susceptibility to disease that occurs with age is primarily the result of an increase in physiologic frailty. Conversely, this frailty leaves us with a decreased ability to withstand stress. I have used the term resilience to refer to this ability to return to homeostasis as a response to a stress. Stress has many varieties including, in addition to the more easily recognizable psychological and environmental types, the stress caused directly or indirectly by disease or, in some cases, the interventions used to combat these same diseases (e.g., surgery, anesthesia, chemotherapy). The age-associated changes in frailty and resilience result in a decrease in the thresholds necessary for disease-specific insults to result in overt pathology. This explains, for example, why a prolonged period being bedridden is only a nuisance to a young individual, but could initiate a deadly cascade in an older adult. Age-related disease burdens can theoretically be alleviated by targeting any or all of these interrelated aspects—frailty, resilience, or the threshold at which insults become overt pathology.
The world as a whole is experiencing what has been called a silver tsunami, in which the convergence of lower birth rates and increased longevity is leading to a profound aging of most societies, including most developing countries. Thus, a concern is that if geroscience is successful, then most chronic diseases will be postponed as a group, life span will increase, and this will exacerbate the problem.[110] Although it is certainly true that life span will increase as a result of these efforts, two fallacies need to be considered: (1) although geroscience aims to postpone all chronic diseases at once, all biomedical research, in all domains, aims to extend life span by curing specific diseases; and (2) it is a mistake to think of these older adults, in current terms, as sick and frail. As discussed, multiple studies in mice and other species have indicated that addressing aging (as opposed to addressing one disease at a time) leads to robust older individuals, not sick ones. In fact, some studies have shown that it is our current approach that will lead to that dreaded outcome. It has been theorized that curing cancer, cardiovascular disease, or both would actually lead to a slight increase in the number of disabled people. This might occur because curing one fatal disease would allow a patient to live longer with other comorbid disabilities and conditions, including sarcopenia, osteoporosis, and sensory loss; although not life-threatening, this would considerably decrease the quality of life. If cured of only one of these diseases, individuals will keep on living with the other limitations until the next life-threatening disease (e.g., Alzheimer disease, diabetes, cancer) does kill them. (Actually, chronic diseases are not cured, they are simply managed.) In contrast, the same calculations have shown that delaying aging even minimally would produce the opposite effect, a decrease in the number of people with disabilities. [111,112] Therefore, under the scenario proposed by geroscience, the “new older adult” would not result in an undue burden on health or pension systems.
In summary, geroscience provides a new platform for thinking about an old problem. What are the molecular and cellular bases for the intersection between aging biology and chronic disease, and conditions that make the former the major risk factor for the latter? It is thought that by understanding this connection, we will be able to address and postpone all diseases and age-related disabilities at once, rather than one at a time, which is the current model.
– by Felipe Sierra, Ph.D. [Editor’s note: this article continues in additional sections.]
Geroscience Article Continues in the 4th Section
- Part 1 of 4: Introduction to the Geroscience essay, and backgrounder on Dr. Sierra.
- Part 2 of 4: The Main Pillars of Research on Aging Biology (the main areas of lifespan-extension research).
- Part 3 of 4: Geroscience (this article).
- Part 4 of 4: A Tentative Look into the Future (how the field of geroscience aims to understand and increase life span.)
Show Us Some Love
- Share this post on social media and help us spread the word– It only takes one click on any of the social media links on this page.
- Follow us on social media – Google+ or Reddit
- Sign up for our email list – We use your email to notify you of new articles. We will not send you spam, and we will not share your email address. You can cancel at any time.
- Tell us what you think of geroscience – Scroll down to enter your comments below.
References Accompanying this Section
(References provided by Dr. Felipe Sierra, accompanying the essay Geroscience.)
2. McCay CM, Crowell MF. Prolonging the life span. Sci Mon 39:405–414, 1934.
3. McCay CM, Crowell MF, Maynard LA. The effect of retarded growth upon the length of life span and upon the ultimate body size. J Nutr 10:63–79, 1935.
13. Kenyon C. The genetics of aging. Nature 464:504–512, 2010.
15. Kennedy BK, Berger SL, Brunet A, et al. Geroscience: linking aging to chronic disease. Cell 159:709–713, 2014.
44. Baur JA, Pearson KJ, Price NL, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 444:337–342, 2006.
97. Niccoli T, Partridge L. Ageing as a risk factor for disease. Curr Biol 22:R741–R752, 2012.
98. Fernández AF, Fraga MF. The effects of the dietary polyphenol resveratrol on human healthy aging. Epigenetics 6:870–874, 2011.
99. Wilkinson JE, Burmeister L, Brooks SV, et al. Rapamycin slows aging in mice. Aging Cell 11:675–682, 2012.
100. Sierra F, Kohanski RA. Geroscience offers a new model for investigating the links between aging biology and susceptibility to aging-related chronic conditions. Public Policy Aging Rep 23:7–9, 2013.
101. Krtolica A, Campisi J. Integrating epithelial cancer, aging stroma and cellular senescence. Adv Gerontol 11:109–116, 2003.
102. Kumar S, Subramanian S. Mutation rates in mammalian genomes. Proc Natl Acad Sci U S A 99:803–808, 2002.
103. Gorbunova V, Seluanov A, Mao Z, et al. Changes in DNA repair during aging. Nucleic Acids Res 35:7466–7474, 2007.
104. Gorbunova V, Seluanov A, Zhang Z, et al. Comparative genetics of longevity and cancer: insights from long-lived rodents. Nat Rev Genet 15:531–540, 2014.
105. Vaidya A, Mao Z, Tian X, et al. Knock-in reporter mice demonstrate that DNA repair by non-homologous end joining declines with age. PLoS Genet 10:e1004511, 2014.
106. Pearson KJ, Baur JA, Lewis KN, et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab 8:157–168, 2008.
107. Martin-Montalvo A, Mercken EM, Mitchell SJ, et al. Metformin improves health span and lifespan in mice. Nat Commun 4:2192, 2013.
108. Harrison DE, Strong R, Sharp ZD, et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460:392–395, 2009.
109. Harrison DE, Strong R, Allison DB, et al. Acarbose, 17-alpha-estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males. Aging Cell 13:273–282, 2014.
110. Mendelson DN, Schwartz WB. The effects of aging and population growth on health care costs. Health Aff 12:119–125, 1993.
111. Miller RA. Extending life: scientific prospects and political obstacles. Milbank Q 80:155–174, 2002.
112. Goldman DP, Cutler D, Rowe JW, et al. Substantial health and economic returns from delayed aging may warrant a new focus for medical research. Health Aff (Millwood) 32:1698–1705, 2013.
References
Cover photo credit: Getty Images / Tommasolizzul.
National Institute on Aging. “Felipe Sierra.” National Institutes of Health. Web, Retrieved 10 Jan 2018. Link to Article.
Felipe Sierra. “Geroscience.” Brocklehurst’s Textbook of Geriatric Medicine and Gerontology, 8th Edition, Authors: Howard Fillit Kenneth Rockwood John B Young. ISBN: 978-0-7020-6185-1
Disclaimer
Diagnosis, Treatment, and Advice: This article is intended for educational and informational purposes only and is not a substitute for qualified, professional medical advice. The information and opinions provided herein should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. Consult a qualified and licensed physician for the diagnosis and treatment of any and all medical conditions. Experimental therapies carry a much higher risk than FDA-approved ones. Call 911, or an equivalent emergency hotline number, for all medical emergencies. As well, consult a licensed, qualified physician before changing your diet, supplement or exercise programs.
Photos, Endorsements, & External Links: This article is not intended to endorse organizations, companies, or their products. Links to external websites, mention or depiction of company names or brands, are intended for illustration only and do not constitute endorsements.

Sorry Brady,
A discussion of aging without mention of mTOR or Blagosklonny is just nonsense.
Hi – I split up Sierra’s essay into four easier-to-read parts. Sierra discusses importance of mTOR in one of the sections. In part 4 of this series, he advocates for rapamycin, metformin and (gulp) resveratrol.