8 Ways Cell Stress Causes Us To Age Prematurely
Summary: Cell stress is a fact of life. The cells in our bodies are subject to stress from toxins such as internally generated free radicals, cigarette smoke, air pollution, and UV light. How our cells deal with stress ultimately controls our health. [Author: Brady Hartman. This article first appeared on the LongevityFacts.com website under the title ‘Eight Ways Your Cells Deal With Stress’ and was updated on Dec 22, 2017]
Your heart rate speeds up. Your muscles tense. Your face may even blush. These are just a few ways your body responds to stress. But stress also can seep into your cells, coming from within the body and without. Internal stressors include free radicals (reactive oxygen species) and toxins produced by pathogens. External stressors include extremes of temperature, mechanical damage, UV light, and environmental toxins such as cigarette smoke and air pollution.
These stressors threaten how your cells function—and ultimately controls your health.
Scientists have learned a great deal about how cells respond to stress, and what it means for your health.
8 Ways Cell Stress Causes Us To Age
1. Telomere Shortening
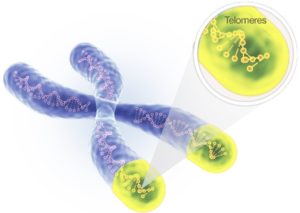
Every cell in your body has clocks called telomeres which limit the number of times a cell can divide. Telomeres look like the plastic tips on the ends of shoelaces and are protective caps on the ends of each chromosome that keep them from fraying.
Telomeres get shorter every time the cell divides, and once it has divided about 50 times, the cell commits suicide through a process called cell death.
Scientists link shortened telomeres to age-related diseases, such as diabetes, arthritis, hypertension, and stroke and as well as the aging process itself. Research also suggests that chronic stress—both cellular and psychological —can significantly shorten telomere length, which in turn causes cells to age and die prematurely.
As a countermeasure, certain cells respond to temporary stressors like psychological stress or infection by upping their production of the enzyme, telomerase, which helps telomeres maintain their length.
2. Epigenetic Changes

Cellular stress can reach deep into our cells’ interiors and alter our epigenome, a part of our genetic code. To make matters worse, these epigenetic changes can be passed on to the next generation.
The epigenome is a structure which rides on top of our genetic code and derives its name as an amalgam of the words ‘epi’ meaning ‘above’ and ‘genome’ for the place where our DNA is kept. The epigenome tells our DNA what to do, where to do it, and when to do it. The epigenome works by a mechanism called the epigenetic program which gives instructions to the DNA using chemical compounds called ‘marks’ that tell the cell which parts of our DNA should be read and which should be ignored. These epigenetic marks are not part of the DNA itself but merely set of overriding instructions.
Researchers have found that these epigenetic changes can influence our health. For example, the toxins produced by cigarette smoking not only damage our cells but damage the epigenome as well.
Epigenetic marks can be passed on from cell to cell as cells divide, and from one generation to the next. For example, a Swedish study found that limited food availability during a man’s lifetime was linked to his grandchildren’s risk of obesity, diabetes, and cardiovascular disease (CVD).
3. When The Mercury Rises
Temperature increases can stress out cells. Warm them just a few degrees, and cellular proteins begin to unravel and stop functioning. If they unravel too much, they tangle up with each other and form a clump that can kill the cell.
To prevent this catastrophe, our cells rely on a set of molecules called molecular chaperones, or heat shock proteins (HSPs), or that work in many different ways. Some HSPs tuck the sticky, carbon-rich regions of unfolded proteins into a small pocket. Other HSPs extend a protective arm around their unfolded neighbors or form into barrel shapes that sequester unraveled proteins, keeping them from becoming entangled with others. Once things cool down, heat shock proteins help their customers refold into proper shapes.
4. Go on a Detox (Response to Toxins)
Toxins entering the cell trigger pathways similar to those involving heat shock proteins, because toxins denature vital cellular proteins in much the same way as heat does. For example, many heavy metals react with sulfhydryl groups stabilizing proteins causing them to change shape. As well, other toxins that cause the release of free radicals can generate unfolded proteins.
5. Adapt By Changing the DNA

Typically, our cells use intricate mechanisms to maintain a stable genetic code. However, during times of stress, they may relax these controls as a coping mechanism. Even though it creates genomic instability, this adaptation allows our cells to adjust to stressful conditions in a short period.
Scientists are searching for ways to exploit this adaptive strategy in the treatment of disease. Scientists feel they could trigger this mechanism to treat cancers, which typically involve cells with extra or missing chromosomes.
Sometimes a cell receives permanent DNA damage because of toxic stress. Obviously, this type of damage isn’t particularly healthy in the long run. Learn more about DNA damage in this article.
6. Slim Down (Autophagy)
Additionally, autophagy is used to counter cell stress, which occurs when the cellular machinery becomes damaged, or proteins stick together in messy clumps called misfolded proteins. In this case, autophagy removes the danger by digesting the culprits. Sometimes autophagy can become impaired, and this condition brings dire consequences.
7. Commit Suicide (Apoptosis)
If all else fails, a cell might kill themselves via a program called apoptosis. Otherwise, if a cell dies in a disorderly fashion, the resulting debris triggers inflammation which can damage healthy cells nearby. In contrast, apoptosis is the orderly kind of cell death.
Apoptosis or programmed cell death occurs as a healthy and controlled part of an organism’s growth and development. During apoptosis, a cell is broken up into small, self-contained pieces, which are easily recycled by white blood cells.
Role of Apoptosis in Disease and Aging
Unfortunately, as we age, our body dials up apoptosis and inflammation. Working together, the two deliver the double whammy that reduces our health and produces our age-related decline. While apoptosis is a necessary process, our bodies lose too many healthy cells to the process, including cells that are still useful.
Sometimes there is too little apoptosis, as in the case of cancer. Tumors possess some mutations that have allowed them to ignore standard cellular signals that act as a check on their growth. Under normal circumstances, damaged cells will undergo apoptosis, but in the case of cancer cells, many have mutations which prevent them from undergoing apoptosis.
On the other hand, sometimes there is too much apoptosis, for example in the case of neurodegenerative diseases such as Parkinson’s or Alzheimer’s. Apoptosis is thought to account for much of the cell death and the progressive loss of neurons.
Triggers of Apoptosis
Researchers don’t yet fully understand how the cell switches from a protective mode to apoptosis, but know it is often kick-started by an accumulation of stress signals, such as low oxygen or damaged DNA. New research suggests that the start of apoptosis has to do with the accumulation of misfolded proteins. Researchers believe stressful conditions, such as exposure to a chemical, produce too many misfolded proteins, which in turn signal apoptosis to start. The cell commits suicide by activating enzymes that degrade proteins, who finish the job by degrading cellular proteins indiscriminately.
How Apoptosis Works
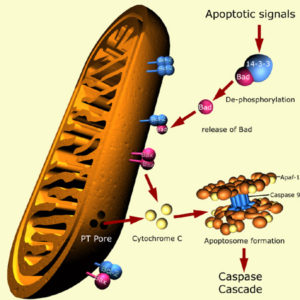
Our mitochondria carry out the death sentence in the apoptosis process.
The initial phases of apoptosis cause the mitochondrial membranes to leak. Once its mitochondria are damaged, a cell is well on its way to the graveyard. These tiny organelles are not the judges who sentence the cell to death but only act as executioners when prompted by external signals.
Apoptosis is a highly regulated process and cannot stop once it has begun. The body initiates apoptosis through one of two pathways, the intrinsic or the extrinsic pathway.
When employing the intrinsic pathway, a cell commits suicide because it senses cell stress. The cell kills itself in the extrinsic pathway because it receives signals from other cells. In both pathways, the cell commits hari-kari by activating caspases or enzymes that degrade proteins. The two pathways both start out by invoking initiator caspases, which in turn activate executioner caspases, who finish the job by degrading cellular proteins indiscriminately.
Because cell death plays a role in some neurological and cardiovascular conditions, scientists are trying to understanding how cells make the life-or-death decision. A better understanding might lead to ways to reduce the damage caused by these diseases.
8. Just Up and Die (Necrosis)
Conventionally, necrosis means a passive, unplanned cell death caused by cellular damage or pathogens, in contrast to orderly, programmed cell death via apoptosis.
Necrosis happens in response to high pressure or high temperature or another strong stressor.
When a cell dies by necrosis, it bursts and releases its guts into the surrounding tissue. It’s a messy way to go, and the mess creates problems for the surrounding healthy tissue and leads to inflammation.
Necrosis can be intentional and actively regulated by the cell and is termed programmed cell death or a programmed form of necrosis. Although programmed forms of necrosis aren’t the unplanned form of death as observed with classical necrosis, these forms are still pretty messy and lead to inflammation. As with passive necrosis, during programmed necrosis, the cell swells until it bursts, spilling its contents into the surrounding space. This messy death and the debris it leaves behind induces inflammation and puts the immune system on alert.
Unfortunately, this inflammation turns against us in chronic diseases. In these conditions, the inflammatory response does not completely remove the primary stimulus. As a result, chronic inflammation ensues that damages our tissues. Scientists have just discovered a link between necrotic forms of cell death and inflammation-related diseases such as diabetes, arthritis, atherosclerosis, and lupus. Learn more in the linked article.
Related article: When Cell Death Goes Bad – Role in Cancer and Inflammation-linked Diseases.
Bottom Line
Our cells are subject to stress from toxins such as cigarette smoke, pollution, and UV light. How our cells deal with the stress ultimately controls our health. When the cells can’t handle the stressors correctly, it can cause damage and inflammation. A well-running stress response mechanism keeps the stress from damaging our cells. Still, stress can produce irreversible damage, including shortened telomeres, epigenetic changes, DNA changes or the loss of tissue, due to cell death. And we can’t afford to lose too many tissues because, unfortunately, some of our tissues do not regrow.
Unfortunately, our cell’s ability to deal with stress declines with age and with specific diseases. Scientists are trying to figure out ways to improve our cell’s stress response, especially when it comes to programmed forms of cell death.
Related Article: Cellular Garb-aging Causes You To Age.
Show Us Some Love
- Share this post on social media and help us spread the word – It only takes one click on any of the social media links on this page.
- Follow us on social media – Google+ | Facebook | Reddit
- Sign up for our email list – We use your email to notify you of new articles. We will not send you spam, and we will not share your email address. You can cancel at any time.
- Tell us what you think – Scroll down to enter your comments below.
References
Cover photo: Eraxion / Getty Images (adapted).
Simone Fulda, Adrienne M. Gorman, Osamu Hori, and Afshin Samali. “Cellular Stress Responses: Cell Survival and Cell Death.” International Journal of Cell Biology, vol. 2010, Article ID 214074, 2010. doi:10.1155/2010/214074. Link to article.
Susmita Kaushik & Ana Maria Cuervo. Proteostasis and aging. Nature Medicine 21, 1406–1415 (2015) doi:10.1038/nm.4001. Published online 08 December 2015
Disclaimer
Diagnosis, Advice, and Treatment: This article is intended for educational and informational purposes only and is not a substitute for qualified, professional medical advice. The information provided in this article should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. Instead, consult a licensed physician for the diagnosis and treatment of any and all medical conditions. Call an emergency hotline such as 911, or an equivalent number, for all medical emergencies. Additionally, consult a licensed physician before changing your diet, supplement or exercise programs. Endorsements, Photos & External Links: This article is not intended to endorse specific organizations, companies, or their products. Links to external websites, mention or depiction of company names or brands, are intended for illustration only and do not constitute endorsements.

10 Replies to “8 Ways Cell Stress Causes Us To Age Prematurely”
Comments are closed.