Can We Stop Mitochondria From Causing Cancer to Grow?
Summary: A new report on mitochondria and cancer shows how our mitochondria help our cancers to grow. With its 37 genes, mitochondria are an attractive druggable target and researchers are looking at this angle to develop powerful cancer cures. This is part 2 of a two-part series. Part 1 of the series on mitochondria is here. [Author: Brady Hartman. This article first appeared on LongevityFacts ]
New evidence is showing that our mitochondria might hold the cure for cancer.
New research shows that the mitochondria within our bodies bend over backward to help cancer cells grow. Once considered an academic backwater, researchers suddenly have a renewed interest in the metabolism of cancer cells and are focusing on the lowly mitochondrion as a potential cure for cancer.
Can Mitochondria Be The Key To Curing Cancer?
Scientists are publishing increasing amounts of evidence showing that cancer-induced changes in our mitochondria contribute to the growth of cancer. As Dr. Dario C Altieri, Head of the Altieri Lab at the Wistar Institute said in a review in the July 2017 British Journal of Cancer,
There is now a resurgent interest in the role of mitochondria in cancer. Long considered controversial or outright unimportant, mitochondrial biology is now increasingly recognised as an important tumour driver.
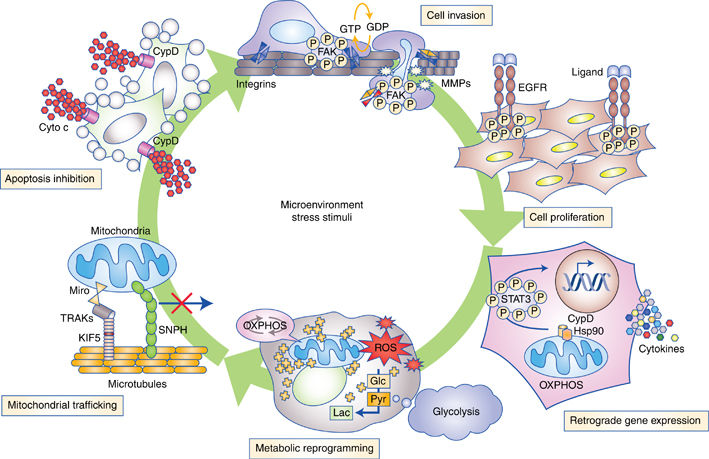
Attractive Druggable Target
Mitochondria are an attractive target for cancer therapy. Unlike the human genome with its 20 to 30 thousand genes, our mitochondria only have 37 genes. A modern bicycle has more moving parts than a mitochondrion.
Compared to the interactions between cancer and the nuclear DNA, the key metabolic interactions between the mitochondria and cancer are few enough that they fit on a wall poster.
Someone already has created such a poster, which is displayed at the end of this report.
By targeting the simple mitochondrion, drug developers have a manageable number of targets to attack. Targeting these mitochondrial genes or their proteins could be a cure for cancer. In a review last year in the journal Molecular Cell, authors Wei-Xing Zong, et al. show the promise of the approach by saying,
Thus, mitochondria play a central and multifunctional role in malignant tumor progression,
and targeting mitochondria provides therapeutic opportunities.
Mitochondrial Kidnapping
What we now know as mitochondria were once bacteria that our cells kidnapped billions of years ago and put to good use. As a result, mitochondria have taken up roles as the powerhouses inside every one of our cells. Depending on the tissue type, each cell has between 100’s or thousands of mitochondria. With their gluttonous appetite for energy, fast-growing cancer cells have even more of the organelles.
Despite the changes over the billion-plus years, our mitochondria still retain a degree of autonomy and possess a different genetic code. Somewhat independent, with their own set of DNA, our mitochondria maintain a comfortable truce with the greater cell, as the two communicate to support vital cellular activity.
Our mitochondria don’t just help healthy cells; they help the cancerous one as well. In fact, a growing body of evidence shows that our mitochondria bend over backward to help cancerous tumors.
Perhaps our mitochondria are exacting revenge because we kidnapped them eons ago?
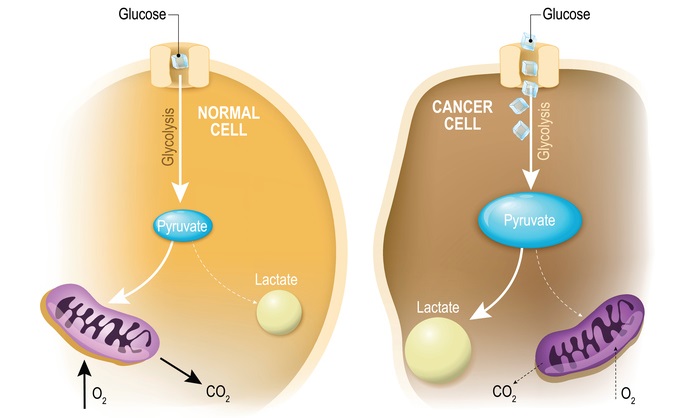
The Warburg Effect
The fact the mitochondrion plays a role in cancer is nothing new. Around the turn of the century, Nobel-prize winner Otto Warburg discovered that cancers use a metabolism different than the one used by ordinary cells, a phenomenon we now call the Warburg effect. Warburg also found that damaged mitochondria helped cancers grow.
Healthy cells combine oxygen and sugar for energy, in a highly efficient process. However, cancer cells use only sugar through the process of fermentation, the anaerobic breakdown of sugar. Cancer cells use fermentation even when oxygen is plentiful, despite the fact that it is far less efficient. Being wasteful consumers of glucose, cancer cells need massive amounts of it, and mitochondria adapt to help the cancer cell burn more of it.
Doctors have long known that sugar feeds cancer and advise their patients to eat a low-carb diet as a way to starve the cancer of its sugar fuel.
The diabetes drug metformin also slashes cancer and is thought to do so by reducing blood sugar levels and targeting the mitochondria. A large systematic review on cancer in metformin users showed that the drug reduced cancer rates by about a third. Sugar feeds cancer cells, and because the metformin reduces blood sugar, it reduces cancer.

The Post-Warburg Effect
The Nobel-prize winner didn’t just come up with the widely-accepted Warburg effect; he also introduced a theory that cancer begins with defective mitochondria, a concept known as the Metabolic Theory of Cancer. While the Metabolic Theory of Cancer hasn’t gained much of a following, the theory has its champions who are labeled the Warburgarians for short and for the sole purpose of this article.
The most outspoken Warburgarian is the self-published writer, Travis Christofferson, author of a book on the subject.
The Warburgarians explain the Metabolic Theory of Cancer as follows: mitochondrial dysfunction starts the cancer ball rolling, and is later followed by nuclear DNA mutations and proliferation. The theory is best summarized by the saying, ‘The origin of cancer, dear Brutus, lies not in our genes, but in our mitochondria.’
The Somatic Mutation Theory, Mitochondria and Cancer
Despite the insistence of the Warburgarians, conventional wisdom backs the Somatic Mutation Theory (SMT) as a cause of cancer. The SMT is used to explain a great many things, however, when the theory explains cancer, it espouses the widely-held belief that cancer starts with a mutation in the nuclear DNA which in turn leads to uncontrolled growth and then proliferation.
No one has entirely verified either of the two theories. However, the Warburgarians like to point to experimental evidence which they claim has dealt the Somatic Mutation Theory a potentially lethal blow.
Attacks on the Somatic Mutation Theory
The attacks discrediting the Somatic Mutation Theory come from two types of experiments chronicled by the another leading Warbugarian, Thomas Seyfried, with the Biology Department at Boston College.
In a 2015 review, Thomas Seyfried related the results of these two types of nuclear cytoplasm transfer (NCT) experiments. In the first type of NCT experiment, when researchers took the nucleus of a cancer cell and transplanted it into a healthy cell, the cell remained healthy. However, in the second NCT experiment, when researchers took the nucleus of a healthy cell and transplanted it into a cancer cell, the cell remained cancerous. The result challenged the Somatic Mutation Theory and the widely-held belief that the essence of cancer is in our chromosomes. Seyfried summarized the belief of the Warburg devotees when he said
“Cancer is widely considered a genetic disease involving nuclear mutations in oncogenes and tumor suppressor genes. This view persists despite the numerous inconsistencies associated with the somatic mutation theory. In contrast to the somatic mutation theory, emerging evidence suggests that cancer is a mitochondrial metabolic disease, according to the original theory of Otto Warburg. The findings are reviewed from nuclear cytoplasm transfer experiments that relate to the origin of cancer. The evidence from these experiments is difficult to reconcile with the somatic mutation theory, but is consistent with the notion that cancer is primarily a mitochondrial metabolic disease.”
Seyfried may have blemished the Somatic Mutation Theory’s role in cancer. However, he and other Warburgarians rely on a faulty premise that goes something like ‘If the Somatic Mutation Theory is wrong, then the Metabolic Theory of Cancer must be right. ‘
Mitochondria and Cancer Compromise
The renowned Douglas C. Wallace tried to settle the academic knife fight between the Warburgarians and the Somatic Mutation Theorists. A geneticist and evolutionary biologist at the Children’s Hospital of Philadelphia, Wallace came to fame having made the landmark discovery that mtDNA are exclusively maternally heritable. Wallace garnered even more attention when he later pioneered the use of human mitochondrial DNA as a molecular marker.
Wallace has not directly contradicted Warburg’s Metabolic Theory of Cancer. Instead, Wallace merely reinterpreted the late, great Otto Warburg by saying,
“Mitochondria were first proposed to be relevant to cancer by Otto Warburg who reported that cancer cells exhibited “aerobic glycolysis.” Although this was originally interpreted as indicating that the function of the mitochondria was defective, we now understand that cancer cells are in an altered metabolic state with increased glycolytic metabolism and the continued use of oxygen.
Rather than ticking the box which supported the Warbugarians, or the other box which supported the Somatic Mutation Theory, it seems that Wallace merely ticked the box which said ‘All of the above.’
“Both somatic and germline mitochondrial DNA mutations have been associated with many types of cancers, and recent data indicate that cancer cells may tolerate mitochondrial DNA mutations for two purposes: they alter cancer cell metabolism and/or proliferation and they enable adaption to a changing environment.”
Wallace wrote the above words in 2012. His words have apparently did not affect the combatants because Christofferson published his book in 2014 and Seyfried published his review in 2015.
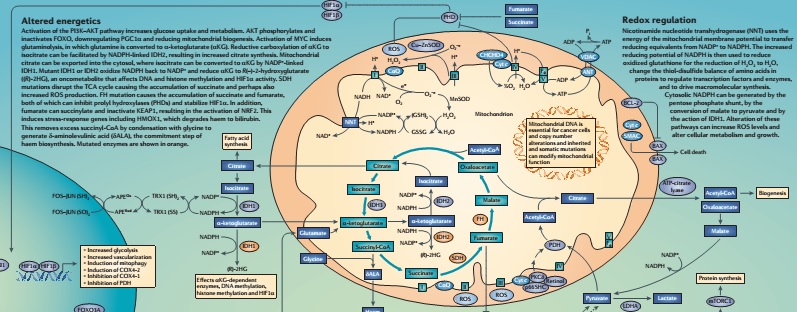
Mitochondria and Cancer Research Move On
Wallace has moved on to bigger and better things, which can only be described as ‘all things mitochondrial.’ Of note, Wallace is joining others in showing how the mitochondria and cancer work together to help cancer grow. In 2012, Wallace summarized that the significant interactions between the mitochondria and cancer. The chemical pathways shared by the mitochondria and cancer are so few, they could fit on a wall poster when Wallace created the poster in 2012. The poster is outdated, as researchers have added new information about the interaction between the mitochondria and cancer. The point of Wallace’s poster is that the mitochondrion, with its measly 37 genes is a relatively straightforward critter to understand from a metabolic point of view.
While the mitochondrion is simple enough, its interactions with cancer are anything but.
Thanks to Altieri, Wallace, and others, researchers are adding to a growing body of evidence which shows that our mitochondria help cancer in many ways. Increasing amounts of evidence show that cancer-induced changes in our mitochondria contribute to the growth of cancer. So far, researchers have found that our mitochondria help tumors grow in all of their stages—initiation, growth, survival, and maintenance. As well, mitochondria are critical to the pathways involved in the growth of cancers, including producing more mitochondria, mitochondrial fusion and division, cell death, regulation of reactive oxygen species (ROS) and other communications inside the cell.
All of these processes represent druggable targets to develop cancer-killing therapies.
Bottom Line
Scientists believe that one cure for cancer lies in attacking the partnership between mitochondria and cancer. Researchers suddenly have a renewed interest in the metabolism and other relationships between the mitochondria and cancer cells. New research shows that the mitochondrion bends over backward to help cancer cells grow.
The lowly mitochondrion is not just hot among cancer researchers, other rapidly growing areas of mitochondrial research include aging and health, evolution and mitochondrial DNA.
As we age, our mitochondrial DNA becomes damaged through mutations that lead to increasing dysfunction. It needn’t be this way, as mitochondria can clone at will at produce healthy DNA. New research is showing fresh insights into the role of mitochondrial DNA in human health and aging, and scientists have made progress reversing the process in animal experiments.
Researchers have made considerable progress in understanding the role that the lowly mitochondria play in aging and health and are looking for ways to interfere with the process.
- Part one of this series: Age-related mitochondrial dysfunction and mutations in mitochondrial DNA are in the spotlight as researchers implicate the tiny organelles in a host of human diseases.
- Researchers discover keys to mitochondrial DNA mutations that play a significant role in diseases.
Show Us Some Love
- One click helps us spread the word – Share this post on social media and help us spread the word. It only takes a click on any of the social media links on this page.
- Follow us on social media – Google+ | Facebook | Reddit
- Sign up for our email list – We use your email to notify you of new articles. We will not send you spam, and we will not share your email address. You can cancel at any time.
- Tell us what you think – Scroll down to enter your comments below.
References
Cover Photo: FatCamera – iStock/Getty Images.
Dario C Altieri. Mitochondria on the move: emerging paradigms of organelle trafficking in tumour plasticity and metastasis. British Journal of Cancer (2017) 117, 301–305. doi:10.1038/bjc.2017.201 www.bjcancer.com. Published online 4 July 2017
Wei-Xing Zong, et al. Mitochondria and Cancer. Molecular Cell, Volume 61, Issue 5, 667 – 676. DOI: http://dx.doi.org/10.1016/j.molcel.2016.02.011
Travis Christofferson, Tripping Over the Truth: The Metabolic Theory of Cancer. Createspace Independent Pub, 8 Oct 2014
Thomas N. Seyfried. Cancer as a mitochondrial metabolic disease. Front Cell Dev Biol. 2015; 3: 43. Published online 2015 Jul 7. doi: 10.3389/fcell.2015.00043. PMCID: PMC4493566
Douglas C. Wallace, Mitochondrial function and cancer, Nature Reviews Cancer. May 2012
Douglas C. Wallace, Nicola McCarthy, Darren Burgess, Lara Crow. Mitochondrial function and cancer. (poster). © 2012 Nature Publishing Group.
Disclaimer
Diagnosis, Treatment, and Advice: This article is intended for educational and informational purposes only and is not a substitute for professional medical advice. The information provided in this report should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. Consult a licensed physician for the diagnosis and treatment of any and all medical conditions. Call 911, or the equivalent emergency hotline number, for all medical emergencies. As well, consult a licensed physician before changing your diet, supplement or exercise programs. Endorsements, Photos & External Links: This article is not intended to endorse companies or other organizations, or their products. Links to external websites, mention or depiction of company names or brands, are intended for illustration only and do not constitute endorsements.

4 Replies to “Can We Stop Mitochondria From Causing Cancer to Grow?”
Comments are closed.