Secrets of these 200-year-old whales who avoid cancer
Summary: Why the bowhead whale lives 200 years and rarely gets cancer. This article is part 2 of a 3-part series, covering the role of telomere length in cancer and part of a new extensive study. Part 1 on the accuracy of telomere length is here, Part 3 on the role of telomere length in chronic diseases is here. [This article first appeared under the title ‘Top Journal Discovers Key to These 200-Year-Old Whales Who Avoid Cancer’ on the website LongevityFacts.com. Author: Brady Hartman. ]
Related: A brief overview of why whales don’t get cancer is in this companion video.
In a comprehensive study published a little more than a week ago in a highly respected journal of the Royal Society of the UK, authors Abraham Aviv and Jerry W. Shay (see author bios here) report on the role of telomere length. In this segment, the authors describe the role of telomere length in protecting us from cancer and explain why whales have low rates of cancer, despite large cell counts, long lives, and relatively short telomeres.
What Are Telomeres?
“I cannot forecast to you the action of telomere length. It is a riddle, wrapped in a mystery, inside an enigma; but perhaps there is a key.” (apologies to W. Churchill)
Telomeres are repeated stretches of DNA that cap the ends of our chromosomes. Telomeres protect our genetic data, keeping the ends of chromosomes from fraying and breaking. Broken chromosomes cause all sorts of trouble and trigger ill health. Telomeres act as a biological clock in our cells. Each time a cell divides, its telomeres get progressively shorter, until it reaches a critically short length. When the telomeres reach this point, the cell can no longer divide and either undergoes cell death or becomes senescent.
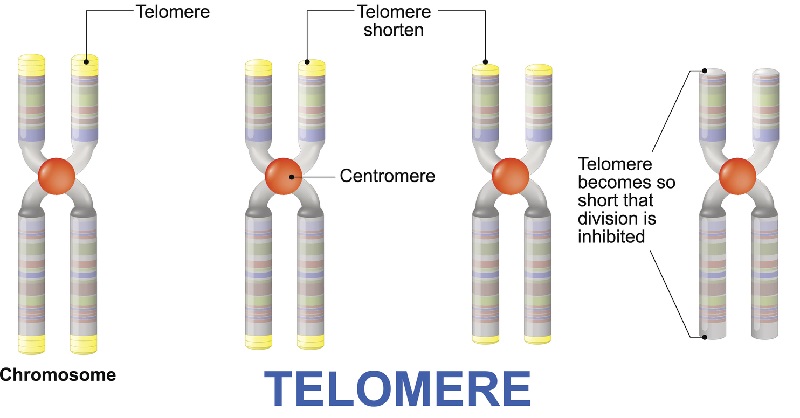
Longevity researchers believe that telomeres are a protective mechanism against cancer. Cancer cells divided without limit and telomeres put a natural limit on a cell’s growth. Unfortunately, cancer cells have mechanisms to never reach this limit. Tumors merely rebuild their telomeres using an enzyme called telomerase.
Richard Peto Faces a Paradox
The authors Aviv and Shay (A+S) point out that evolution has driven telomere dynamics and that cancer resistance mechanisms in large and long-lived mammals explain Peto’s Paradox.
Peto’s Paradox – named after its discoverer, Richard Peto – is the observation, that at the level of a species, the incidence of cancer does not correlate with the number of cells in an organism. For example, the rate of cancer in humans is much higher than that in whales. This is despite the fact that a whale has many more cells than a smaller human. If the probability of cancer were constant across cells, one would expect whales to have a higher incidence of cancer than humans.
Evolution is primarily driven by the interaction between environmental factors and the development of cancer, say A+S, who quote Peto’s Paradox. When compared to humans, large mammals such as elephants and whales have considerably more dividing cells. The difference is even higher when comparing these large animals with mice that are routinely studied in laboratory settings.
For example, consider the differences between an adult mouse and an elephant. The former weighs thirty grams and lives 2–3 years and the latter weighs six tons and lives 60 years. The elephant to mouse ratio is 200,000 for body weight and 60 for lifespan, respectively. Cells need to divide far more often to grow and maintain an elephant’s body when compared to that of a mouse. Since DNA doesn’t always replicate correctly, the overall mutational burden and cancer risk of the elephant would be more than a million-fold higher than that of the mouse. However, Peto pointed out that cancer risk does not always scale with body mass.
Why the Bowhead Whale Lives 200 Years
When compared to small animals, humans are long-lived and share traits with the elephant, and the bowhead whale – such as repressed telomerase activity and short telomeres. According to the prevailing theory, short telomeres and repressed telomerase should limit the ability of cells to divide and inhibit cancer.
Aviv and Shay point out that recent studies found that the elephant and bowhead whale have increased copies of “specific genes that may reduce cancer risk.” A bowhead whale may live up to 200 years making it the longest known living mammal on earth. Asian Elephants reach a maximum lifespan of about 86 years. A+S point out that genome-wide DNA sequencing in elephants detected increased copies of cancer-fighting TP53-related gene sequences and some of these produce functional proteins. The authors add
“Thus, the elephant’s resistance to cancer may be in part due to extra copies of a powerful tumour suppressor gene. While the bowhead whale shows no increase in TP53 dosage, its DNA displays increased variants or copies of DNA damage repair genes (mutations in ERCC1 and PCNA and FEN1 duplications) that may account for augmented cancer resistance.”
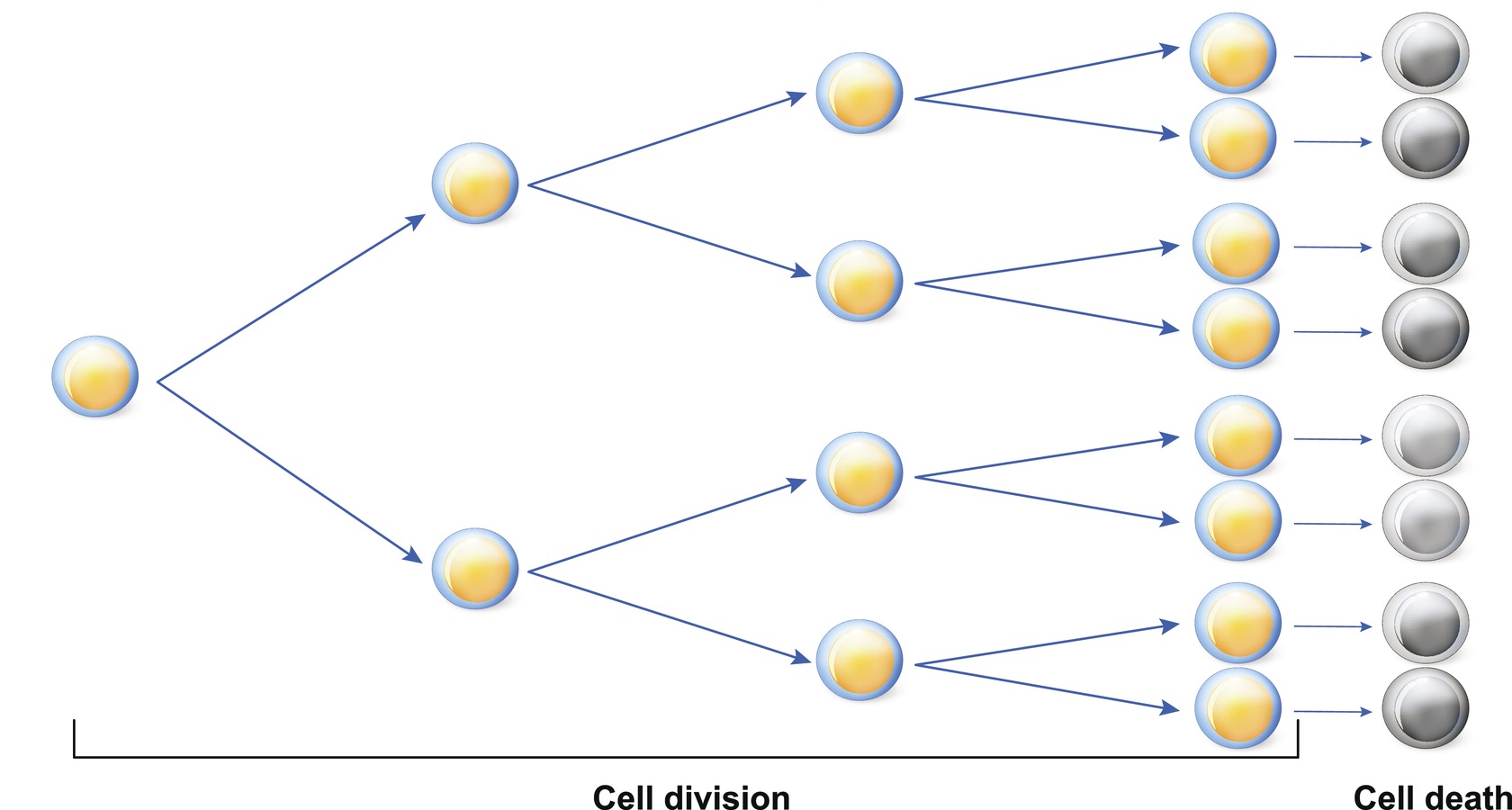
Telomerase Rebuilds Telomere Length
Many different types of proteins are involved in telomere up-keep, the most important one is the enzyme telomerase. Cancers and stem cells both use telomerase to stay immortal. As a cell divides, it makes two copies of its chromosomes. This process gets messy and can shorten the telomeres. Telomerase rebuilds the shortened ends.
Our bodies have two types of cells: stem cells which are immortal, and regular cells, called somatic cells which have a limited lifespan. Immortal cells use telomerase, while somatic cells don’t.
Embryo cells and stem cells are involved in growth and repair. Because they need to be immortal to do their jobs, they rebuild their telomeres using telomerase. Telomerase keeps their telomere length the same despite repeated cell divisions.
Unfortunately, cancer cells also use telomerase to keep their telomere length constant and remain immortal.
Of Mice and Men, Telomerase and Cancer
On the other hand, a group of scientists led by Bruno Bernardes de Jesus experimented using telomerase to extend the lifespan of mice, without causing cancer.
In this experiment, researchers inserted telomerase into mice with the aid of a viral vector. In investigators divided the mice into two sets: adult mice and elderly ones, and treated them with telomerase. At the conclusion of the experiment, the scientists noticed that the telomerase-treated adult mice lived 24 percent longer while the older rodents lived only 13 percent longer. Notably, the treated mice did not have the side effect of increased cancer.
However, these results are ambiguous, according to evidence presented by Aviv and Shay.
A+S explore the idea that short telomeres protect against cancer. Using an extensive series of mammals, but mostly mice, scientists have experimentally tested the idea that repressed telomerase activity and short telomeres are cancer protection mechanisms and the concept remains a valid explanation.
In these experiments, researchers genetically engineered mice to knockout their ability to produce telomerase, the enzyme that rebuilds and lengthens telomeres. A+S say that telomerase knockout mouse cells immortalize at the same frequencies as normal mouse cells – about a ten million-fold greater rate than cells in humans.
However, even the shortest mouse telomeres are longer than the longest telomeres of humans. A+S suggest that mice and many other short-lived small mammals do not use telomere dynamics to protect against cancer. Instead, say the authors, “their long repetitive G-rich telomere sequences might have evolved to serve as a buffer that reduces the hydroxyl radical damage to critical coding genes.” Aviv and Shay add,
“Moreover, genes near telomere ends may be regulated by TL-dependent [telomere length dependent] chromatin interactions, a phenomenon known as telomere position effects over long distances (TPE-OLD). Genes such as human ISG15 and TERT might become dysregulated with progressive telomere shortening. These findings might have implications for how cells turn off telomerase when telomeres reach an optimal length during fetal development and how most cancer cells reactivate telomerase when telomeres become short. Thus, replicative senescence due to short telomeres may paradoxically augment cancer risk through telomerase de-repression. We note that such a phenomenon would survive selection because it principally occurs when telomeres are short, i.e., beyond the reproductive period when the force of evolution wanes.”
Telomere Length and Cellular Senescence
Telomeres act as a limiter to keep the cell from dividing too much. Every time a cell divides, its telomeres get shorter. Once a cell has replicated about 50 times, the cell becomes senescent, and eventually commits suicide via cell death. Not all senescent cells commit cell death, however, and many hang around like the walking dead, creating havoc in the surrounding tissues.
Most scientists say that cellular senescence is a protective mechanism against cancer. However, Aviv and Shay say this is not clear-cut. Senescent cells secrete bucket loads of inflammatory molecules called senescence-associated secretory pathway (SASP) proteins. SASP molecules can lead to dysfunctional tissues and provide an environment that favors cancer. Studies show that clearing out these senescent cells improves tissue function in mice, making them healthier in the process. The authors add that when taken together, these observations indicate that senescent cells
“might bring about both tissue degeneration and cancer susceptibility through mechanisms that are telomere dependent and independent.”
TA-65 and Telomere Length
Some scientists believe that we can live longer, healthier lives if we can restore our telomere length, and are developing treatments to accomplish this. One such drug is TA-65 – a naturally-derived telomerase activator.
Researchers are studying TA-65 to see if it reduces the level of senescent cells. TA-65 seems to increase telomerase levels in mouse tissue but does not have any effect on their lifespan.
This result, combined with the fact that mice with a shortage of telomerase do not live shorter lives than wild mice adds credence to the suggestion of Aviv and Shay that mice do not use telomere dynamics to protect against cancer, and have other protective mechanisms. It also shows that mice are a lousy model when it comes to telomere length and cancer in humans.
Telomere Length and Risk of Cancer
In part 3 of these series, Aviv and Shay point out that the risk of cancer due to long telomeres is small, and this might be because the optimal telomere length strikes a balance between cancer and cardiovascular disease.
Series Articles on Telomere Length
- Part 1 on telomere length accuracy is here.
- Part 3 on the function of telomere length in chronic diseases is here.
- This article is featured in the best of telomere reports series.
Show Us Some Love
- Share this post on social media and help us spread the word– It only takes one click on any of the social media links on this page.
- Follow us on social media – Google+ or Reddit
- Sign up for our email list – We use your email to notify you of new articles. We will not send you spam, and we will not share your email address. You can cancel at any time.
- Tell us what you think – Scroll down to enter your comments below.
References
Cover photo: Bowhead whales have short telomere length. Credit: Corey Ford / Getty Images (iStock)
Abraham Aviv, Jerry W. Shay. “Reflections on telomere dynamics and ageing-related diseases in humans.” Philosophical Transactions of The Royal Society B Biological Sciences, Published 15 January 2018.DOI: 10.1098/rstb.2016.0436. Link to Article.
Bernardes de Jesus, Bruno et al. “Telomerase Gene Therapy in Adult and Old Mice Delays Aging and Increases Longevity without Increasing Cancer.” EMBO Molecular Medicine 4.8 (2012): 691–704. PMC. Web. 24 Jan. 2018. Link to Article.
Disclaimer
Diagnosis, Treatment, and Advice: This article is intended for educational and informational purposes only and is not a substitute for qualified, professional medical advice. The information and opinions provided herein should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. Consult a qualified and licensed physician for the diagnosis and treatment of any and all medical conditions. Experimental therapies carry a much higher risk than FDA-approved ones. Call 911, or an equivalent emergency hotline number, for all medical emergencies. As well, consult a licensed, qualified physician before changing your diet, supplement or exercise programs.
Photos, Endorsements, & External Links: This article is not intended to endorse organizations, companies, or their products. Links to external websites, mention or depiction of company names or brands, are intended for illustration only and do not constitute endorsements.

Why would anybody think Peto’s paradox is a paradox. Basic concept of cancer theory is body has evolved very many extremely effective means to prevent cancer. Mice die of cancer at age 3. Humans generally make it to 80’s before cancer big risk. Without getting into any specifics, cancer is a basically failure of good house keeping.
yes – good point.