Researchers crack secret code to genetic changes causing our cancer
Summary: Researchers at the University of California discovered a key way that cancer manipulates the genetic code using DNA methylation that has important implications for the treatment of cancers. [This article first appeared on the website LongevityFacts.com. Author: Brady Hartman. ]
Up until now, scientists haven’t fully understood how DNA methylation causes changes in our genetic code that enable cancer to thrive.
Now, a team led by associate professor Jikui Song at the University of California Riverside have deciphered the crystal structure of an enzyme that plays a crucial role in DNA methylation that allows tumors to survive and grow.
Dr. Song explained that this finding would enable scientists to master the DNA methylation process, which plays an essential role in the epigenetics of aging and cancer.
“It especially has important implications in cancer therapy in the long term.” – Dr. Jikui Song.

The discovery by the UC Riverside team opens the door to new approaches for future cancer treatments. When gene activity runs amok, as often occurs as the result of DNA damage, it can lead to cancer. Researchers have long known that tumors grow by making epigenetic modifications.
Up until the finding by Song’s team at UCR, scientists have not fully resolved how DNA methylation is faithfully regulated, and how it can become defective. The results of the study appear in the February 7 issue of the prestigious journal Nature (paywalled.)
The finding has particular importance, as researchers are developing a technique called regenerative programming – the use of Yamanaka factors to control the epigenetic program that controls our human genetic code.
The Importance of DNA Methylation
DNA contains the blueprints of our bodies. Every cell in our body uses these genetic instructions to learn what it must become and what function it must perform. Throughout our entire lifespan, this genetic information has to be read correctly to ensure that genes are active in the right cells and at the right time. If these genetic processes are defective, cells can acquire the wrong identity – which could ultimately lead to cancer.
However, the program that determines which of our genes are switched on or off as the cell develops does not depend solely on DNA. Epigenetic marks also affect the program. Marks on our DNA, known as DNA methylation, act as molecular switches that regulate gene activity to coordinate the cell’s specialization within our bodies.
DNA methylation alters the way in which are genetic code is read and interpreted. Methylation can change the activity of a segment of DNA without changing its sequence. During the process of DNA methylation, methyl groups are added to the DNA molecule, which in turn alters gene expression.
DNA methylation is a natural process that is essential for healthy development of our bodies. DNA methylation is a fundamental cellular mechanism that critically influences our bodies as it regulates genetic stability and cell differentiation. On the other hand, researchers link errors in methylation with the aging process and various human diseases, most importantly cancer.
DNA Methylation and Cancer
The UCR finding will help researchers battle cancer. The consequences of defects in the DNA methylation process are well known: In many types of cancer, methylation is performed in the wrong place, leading to genes being read incorrectly.
Science has known for some time that cancers use DNA methylation to silence many tumor suppressor genes, and other scientists are attempting to re-express these cancer-fighting genes by inhibiting the DNMT enzymes. The UCR discovery will aid in this avenue of cancer treatment.
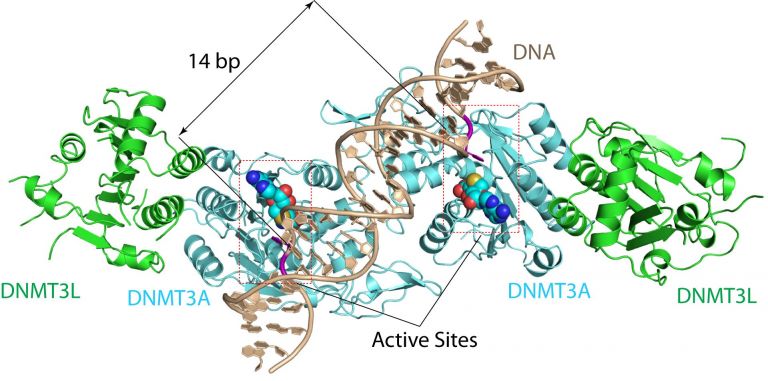
UCR’s DNA Methylation Discovery
There are two types of DNA methylation; maintenance methylation and de novo methylation – a term describing the methylation that occurs during germ cell development and early embryonic development.
De novo DNA methylation is carried out by the enzymes DNMT3A and DNMT3B. Researchers have had a hard time understanding how de novo DNA methylation works, because, until recently, scientists have not known the structures of these enzymes.
The team led by Dr. Song at UC Riverside solved the crystal structure for substrate-bound DNMT3A. This breakthrough shows how the DNMT3A enzyme recognizes and methylates its targets known as substrates. This is a significant finding to further our understanding of de novo DNA methylation. A substrate is a substance on which an enzyme acts, and in this case, the substrate of the DNMT3A enzyme is our genetic code. As Jikui Song says,
“The structure reveals that DNMT3A molecules attack two substrate sites adjacent to each other on the same DNA molecule,” “This now offers us a much clearer view on how de novo DNA methylation takes place.”
Moreover, the finding will aid in the fight against cancer, as Dr. Song says,
“Our work presents the first structural view of de novo DNA methylation and presents a model for how some DNMT3A mutations contribute to cancers, such as acute myeloid leukemia. This study should provide important insights into the function of DNMT3B as well.”
The DNMT3A structure cracked by professor Song’s team explains why DNA methylation predominantly occurs at locations on our genome called CpG dinucleotides— places in the DNA where cytosine nucleotides lie next to guanidine nucleotides.
“Before our study, why mammalian DNA methylation mostly occurs at the CpG sites was not understood, and our understanding of de novo DNA methylation was purely based on computational modeling, which cannot reliably explain how DNMT3A works,” professor Song said. “Just how DNMT3A succeeded in binding to its substrate was not understood either. Our structure for DNMT3A-DNA complex addresses all these concerns, offering a far better understanding of how specific DNA methylation patterns are generated.”
Significance of the DNA Methylation Discovery
Up until this finding, the study of the DNMT3A structure with substrates has been hindered by scientist’s difficulty in producing a stable enzyme-substrate complex.
“To overcome this challenge, we successfully developed a method to trap the reaction intermediate of DNMT3A-substrate complex, and solved the structure by X-ray crystallography,” professor Song said.
Dr. Song was joined in the study by other researchers at the University of California at Riverside as well as researchers at the University of North Carolina at Chapel Hill. Professor Song’s group solved the crystal structure, and the UNC-Chapel Hill team performed the genomic DNA methylation analysis.
Related: Researchers at CWRU inhibit the spread of cancer with a new technique.
Show Us Some Love
- Share this post on social media and help us spread the word– It only takes one click on any of the social media links on this page.
- Follow us on social media – Google+ or Reddit
- Sign up for our email list – We use your email to notify you of new articles. We will not send you spam, and we will not share your email address. You can cancel at any time.
- Tell us what you think – Scroll down to enter your comments below.
References
Cover photo: DNA methylation. Credit: Christoph Bock, Max Planck Institute for Informatics – Own work/ CC BY-SA 3.0 (adapted).
Iqbal Pittalwala. “Scientists crack structure of enzyme complex linked to cancer.” University of California – Riverside. February 7, 2018. Link to the UCR press release.
Zhi-Min Zhang et al. “Structural basis for DNMT3A-mediated de novo DNA methylation.” Nature (2018). DOI: 10.1038/nature25477. Link to the article in Nature. (paywalled).
Disclaimer
Diagnosis, Treatment, and Advice: This article is intended for educational and informational purposes only and is not a substitute for qualified, professional medical advice. The information and opinions provided herein should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. Consult a qualified and licensed physician for the diagnosis and treatment of any and all medical conditions. Experimental therapies carry a much higher risk than FDA-approved ones. Call 911, or an equivalent emergency hotline number, for all medical emergencies. As well, consult a licensed, qualified physician before changing your diet, supplement or exercise programs.
Photos, Endorsements, & External Links: This article is not intended to endorse organizations, companies, or their products. Links to external websites, mention or depiction of company names or brands, are intended for illustration only and do not constitute endorsements.