Gene Therapy for Blindness May Soon Be Reality
Summary: The success of a novel gene therapy for blindness caused by a genetic mutation paves the way for gene therapies which treat other forms of blindness as well as genetic therapies which treat other diseases. [This article first appeared on LongevityFacts on Nov 11, 2017, and has been updated on Dec 23, 2017, to reflect the fact that Luxurna’s gene therapy for inherited blindness received FDA approval recently.]
After being treated with a novel gene therapy for blindness, patients who had lost their sight due to a genetic retinal disease could see well enough to navigate a maze, according to research presented today at the 2017 Annual Meeting of the American Academy of Ophthalmology (AAO 2017).
Novel Gene Therapy for Blindness
Patients in the study had a form of blindness called Leber congenital amaurosis (LCA), an inherited degenerative retinal disease characterized by severe loss of vision at birth. This novel gene therapy for blindness is currently under review by the FDA for potential approval. There are no other FDA-approved treatments available for inherited retinal diseases.
Stephen R. Russell, M.D. – an Ophthalmologist with the University of Iowa – is one of the lead researchers for this trailblazing treatment. Data from the first randomized and controlled phase III study showed that 27 of 29 patients treated with the gene therapy experienced meaningful improvements in their vision, enough that they could navigate a maze in low to moderate light. The treated patients also showed improvement in light sensitivity and peripheral vision, two common visual deficits experienced by these patients.
How the Gene Therapy for Blindness Works
Leber congenital amaurosis is a rare disorder, affecting about 1 in 80,000 people. The condition can be caused by any combination of 19 different genes. The most common cause is a defect in the RPE65 gene which produces a protein of the same name called RPE65. This protein helps the cells that nourish the retina – called the retinal pigment epithelial cells or RPE – to process vitamin A. The RPE cells of patients who have this mutation do not produce enough vitamin A, an essential nutrient that allows visual proteins to sense light. Fixing the condition requires delivering a DNA segment with a healthy copy of the RPE65 gene packaged in a vector made from a virus.
The treatment, called voretigene neparvovec (Brand name: Luxturna, from Spark Therapeutics), uses a harmless virus that is genetically modified to carry a healthy version of the gene into the retina. Ophthalmologists inject billions of these modified viruses into both of the patient’s eyes.
Luxturna’s gene therapy does not restore normal vision. However, it allows patients to see shapes and light and enables them to walk around without a cane or a guide dog. Researchers are unsure how long the treatment will last. So far, most of the treated patients have maintained their vision for two years.
Earlier Trials Testing Gene Therapy for Blindness
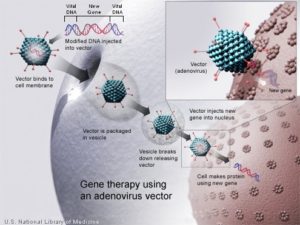
More than 200 patients with Leber congenital amaurosis have participated in trials of gene therapy for blindness since 2007.
The first trial was supported by the National Eye Institute (NEI) and began in 2007 and used a modified adeno-associated viral vector to deliver the healthy RPE65 gene to the retina.
In 2008, the researchers published the results of their three-month follow-up in the Proceedings of the National Academy of Sciences and the journal Human Gene Therapy.
Dr. Samuel Jacobson, one of the researchers attached to the earlier trial, related what the patients reported after receiving the gene therapy,
“So what do they notice, well, generally speaking within a week, they will tell you that they see things brighter. They will tell you that they see things. Some are more specific observers and others are less specific. Some will tell you where exactly where they see it brighter. One person calls it a headlight. The headlight got turned on on day five and it just hasn’t turned off. That’s a really interesting headlight. I mean it’s not a headlight that’s there without vision, stimulation but there’s a view of the world through that headlight that wasn’t there before.”
In August 2009, researchers published their one-year results in the same journal, Human Gene Therapy, and in a letter to the editor of the New England Journal of Medicine. The researchers reported that all three of the treated patients remained healthy and maintained previous visual gains. One patient also experienced a visual improvement that helped her perform daily tasks. Dr. Redmond spoke of the significance of the favorable results, saying
“The LCA gene therapy trial is highly significant to the field of vision in that it was proof of principle that genetic blindness could be reversed. It provides a template, for future work in gene therapy, not only of the eye, but also of other systems in the body.”
FDA Approval Expected
The latest phase III study presented at the AAO 2017 confirms the efficacy of this gene therapy for blindness. No other gene therapy has gotten this close to FDA approval for retinal disease or any other eye disease. In October of this year, an advisory committee to the FDA unanimously endorsed the treatment. The FDA is not obligated to follow the recommendations of its advisory committees, but it usually does. Observers expect the FDA to make its decision on this gene therapy for blindness no later than January 2018.
Gene Therapy for Other Forms of Vision Loss
FDA approval of Luxturna could open the door for other gene therapies that could eventually treat the more than 225 genetic defects known to cause blindness. For example, gene therapy could be used to treat retinitis pigmentosa, an inherited retinal disease caused by a genetic defect. Moreover, gene therapy could potentially provide key proteins needed to restore vision in more common forms of vision loss, such as age-related macular degeneration (AMD).
Bottom Line
Years of research using gene therapy are starting to bear fruit. Unfortunately, not all experiments are carefully controlled, such as the one presented at AAO 2017. For example, one man is self-experimenting with gene therapy to rejuvenate his aging body. The successful trial in treating blindness is only the beginning of the application of gene therapy. Researchers hope that gene therapy can be used to treat other forms of blindness, and eventually other diseases.
Related Articles on Gene Therapy and Gene Editing
- Learn more about gene editing in this video on the Future of Medicine.
- (video) CRISPR 2.0 – New Ways to Edit Genes in Our Bodies – a video that explains CRISPR.
- MIT researchers discover a Revolutionary CRISPR Gene Editing with Nanoparticles.
- Discover how old human cells became rejuvenated with the compound resveratrol.
- Microsoft’s Bill Gates to Genetically Engineer Laser Lit Mosquitos
- British physicians say they have achieved “mind-blowing” results using gene therapy to rid people of hemophilia A.
Show Us Some Love
- One click helps us spread the word – Share this post on with your friends on social media. It only takes one click on any of the social media links on this page.
- Follow us on social media – For more articles, follow us on Google+ | Facebook | Reddit
- Sign up for our email list – We use your email to notify you of new articles. We won’t send you spam, and we won’t share your email address. You can cancel at any time.
- Tell us what you think – Please scroll down to enter your comments.
References
American Academy of Ophthalmology. Genetic treatment for blindness may soon be reality. [Press release]. American Academy of Ophthalmology. November 11, 2017. Link to Article.
National Eye Institute. The Research Road: Gene Therapy for Leber Congenital Amaurosis. National Eye Institute. (Online publication). Web. Retrieved Nov 11, 2017. Link to Article.
Disclaimer
Diagnosis, Treatment, and Advice: This article is intended for educational and informational purposes only and is not a substitute for qualified, professional medical advice. The information and opinions provided herein should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. Experimental therapies carry a much higher risk than FDA-approved ones. Consult a licensed and qualified physician for the diagnosis and treatment of any and all medical conditions. Call 911, or an equivalent emergency hotline number, for all medical emergencies. As well, consult a licensed, qualified physician before changing your diet, supplement or exercise programs. Photos, Endorsements, & External Links: This article is not intended to endorse organizations, companies, or their products. Links to external websites, mention or depiction of company names or brands, are intended for illustration only and do not constitute endorsements.

5 Replies to “Gene Therapy for Blindness May Soon Be Reality”
Comments are closed.